Vicero's VCR-036: A Breakthrough Dual-Targeting VHH Antibody for Cancer Immunotherapy
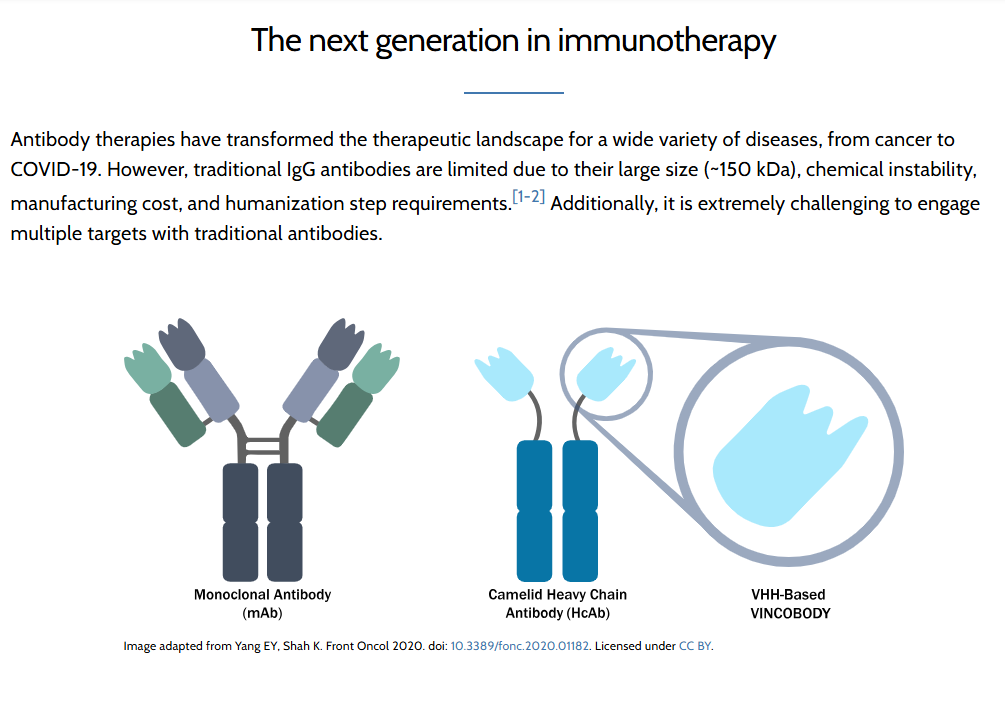
The VCR-036, a bispecific VHH antibody, has been designed to overcome the limitations of traditional immuno-oncology treatments, addressing key issues of toxicity and efficacy seen with existing therapies.
Key Findings from Preclinical Studies:
Improved Safety Profile: VCR-036 demonstrated an impressive 100% survival rate in preclinical models, in contrast to the 50% survival rate achieved with standard combination therapies, such as PD-1 and CTLA-4 monoclonal antibodies.
Complete Tumor Regression: A 5 mg/kg dose of VCR-036 led to complete tumor regression, with sustained remission observed after tumor rechallenge.
Optimized Dosing Schedule: Pharmacokinetic studies suggest that VCR-036 could be dosed at a 1 mg flat subcutaneous dose every six weeks, offering patients a more convenient treatment regimen compared to traditional therapies.
Fewer Immune-Related Adverse Events: The VCR-036 VHH antibody showed a significant reduction in immune-related adverse events compared to combination treatments like pembrolizumab and ipilimumab, potentially minimizing treatment-related side effects.
A New Era in Cancer Immunotherapy:
“Our VINCOBODY platform allows us to develop innovative therapies like VCR-036, which offers the efficacy of dual checkpoint blockade while mitigating the toxicity often associated with traditional monoclonal antibodies,” said Vikram Kansra, PhD, Founder and CEO of Vicero. “The promising preclinical data for VCR-036 underscores the potential of our approach to expand the therapeutic window for patients with solid tumors.”
By utilizing single-domain antibodies (VHHs), Vicero aims to deliver more precise and safer treatments for cancer patients. VCR-036 is positioned to redefine the standard of care in immuno-oncology by addressing significant challenges in combination immunotherapy.
Transforming Cancer Care with Dual-Target VHH Antibodies:
“We are excited about the potential of VCR-036 to meet the growing demand for more effective and safer cancer treatments,” said Alison Finger, Chief Operating Officer of Vicero. “The dual-targeting nature of VCR-036 combined with its six-week subcutaneous dosing offers meaningful advantages for patients, reducing treatment burden while increasing convenience.”
With its breakthrough data, VCR-036 is poised to become a key player in the large checkpoint inhibitor market, offering a novel solution for cancer patients while pushing the boundaries of VHH antibody technology. Vicero is committed to advancing this promising therapy to clinical trials and ultimately providing better outcomes for those with solid tumors.
News Source: BUSINESS WIRE